Lead (Pb) ranks as a major anthropogenic pollutant because it is used extensively by industry, with mining industry and fossil fuel burning accounting for significant levels of lead released into the environment. Various industrial activities including the production of batteries, ammunition, plumbing, alloys, lead crystal glassware, shields for X-ray equipment and nuclear reactors employ Pb as a raw material. Lead ion, Pb(II), is highly toxic to all biological systems, inhibiting cell growth with detrimental effects on protein synthesis, and leading to alteration of the osmotic balance, enzyme inhibition, nucleic acid damage, disruption of membrane functions and oxidative phosphorylation.
This topic explores the complex chemistry of Pb vis-à-vis bacteria in the framework of resource recovery and bioremediation of industrial effluents.
Collaborators: Larry Barton (University of New Mexico, USA) and Mihaly Posfai (University Pannonia, Veszprem, Hungary).
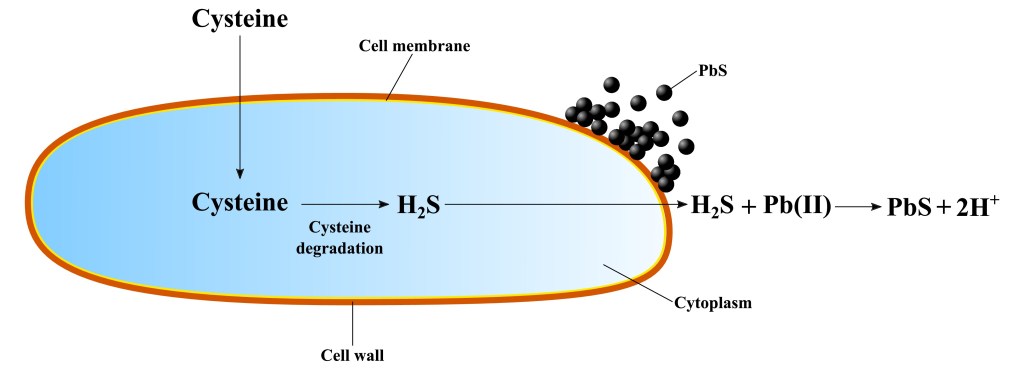
The article demonstrates for the first time the use of cysteine by a Bacillus cereus strain to detoxify lead (Pb) by forming insoluble PbS (galena) minerals. Apart from its fundamental significance such as understanding the biogeochemistry of Pb in anthropogenic and natural settings, this work may open the possibility to recycle this valuable metal from high-strength industrial effluents. PbS is currently the main source of Pb, being mined and processed through energy-intensive and environmental-sensitive pyrometallurgical processes.
Staicu LC, Wojtowicz PJ, Pósfai M, Pekker P, Gorecki A, Jordan FL, Barton LL. (2020) “PbS biomineralization using cysteine: Bacillus cereus and the sulfur rush”. FEMS Microbiol Ecol 9(96) (read)